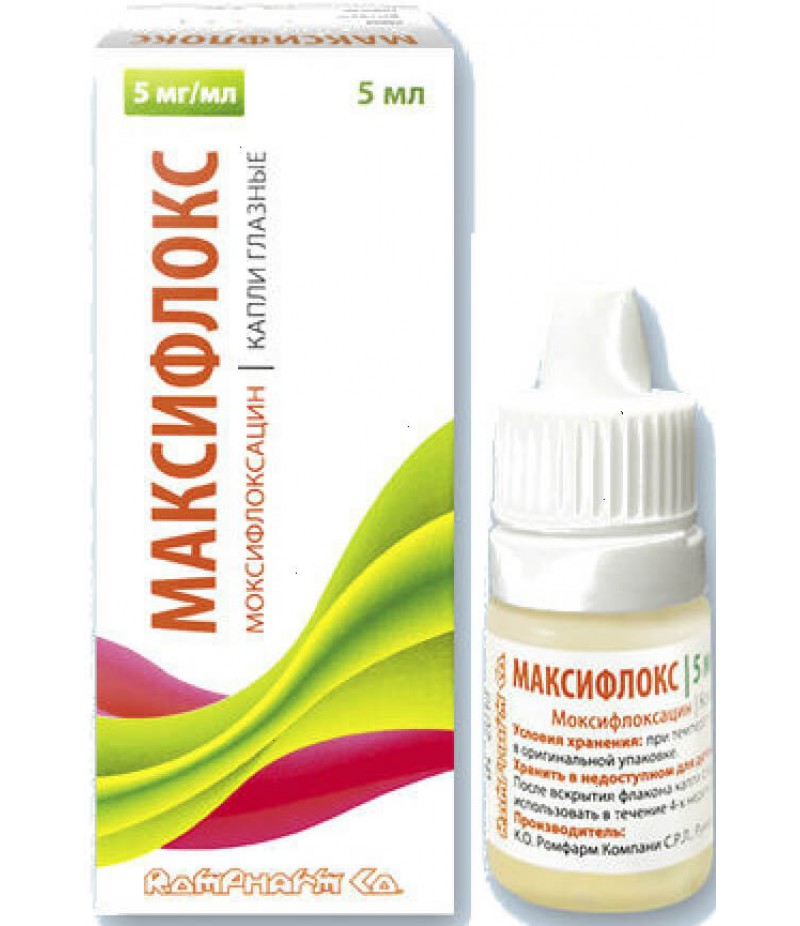
Maxiflox drops 5mg/ml 5ml
- $9.59
- 3 or more $9.30
- Availability:In Stock
Maxiflox user manualReed more and buy Maxiflox on this pageCompositionper 1 ml of the drug:active substance: moxifloxacin hydrochloride 5.450 mg in terms of moxifloxacin 5,000 mg;excipients: boric acid - 3,000 mg, sodium chloride ..
Tags: drops
Maxiflox user manual
Reed more and buy Maxiflox on this page
Composition
per 1 ml of the drug:
active substance: moxifloxacin hydrochloride 5.450 mg in terms of moxifloxacin 5,000 mg;
excipients: boric acid - 3,000 mg, sodium chloride -6,500 mg, 1 M hydrochloric acid / 1 M sodium hydroxide solution to pH = 6.7 - 7.0, purified water - up to 1,000 ml.
Description
Greenish yellow, clear solution.
Pharmacotherapeutic group
Antimicrobial agent - fluoroquinolone
ATX Code:
S01AE07
Pharmacological properties of Maxiflox
Pharmacodynamics
Mechanism of action
Moxifloxacin is a fluoroquinolone antibacterial preparation of the IV generation, inhibits DNA gyrase and topoisomerase IV, which in the bacterial cell performs DNA replication, recombination and repair.
Mechanisms of development of resistance
Resistance to antibiotics of the fluoroquinolone series, including moxifloxacin, develops through chromosomal mutations in genes encoding DNA-gyrase and topoisomerase IV. In Gram-negative bacteria, resistance to moxifloxacin is associated with mutations in a system of multiple resistance to antibiotics and a system of resistance to quinolones. The development of resistance is also associated with the expression of efflux proteins and inactivating enzymes.
Cross-resistance with macrolides, aminoglycosides and tetracyclines is not expected due to differences in the mechanism of action. The development of resistance may have significant geographical differences, and also vary significantly at different periods of time, so before starting therapy it is necessary to obtain information about the resistance of microorganisms in a particular locality, which is of particular importance in the treatment of severe infections.
Moxifloxacin is active against most strains of microorganisms (both invitro and b invivo):
Gram-positive bacteria
Corynebacteriumspp., Including Corynebacterium diphtheriae;
Micrococcusluteus (including strains not susceptible to erythromycin, gentamicin, tetracycline, and / or trimethoprim);
Staphylococcusaureus (including strains insensitive to methicillin, erythromycin, gentamicin, ofloxacin, tetracycline, and / or trimethoprim)
Staphylococcus epidermidis (including strains insensitive to methicillin, erythromycin, gentamicin, ofloxacin, tetracycline and / or trimethoprim);
Staphylococcusaemolyticus (including strains insensitive to methicillin, erythromycin, erythromycin, gentamicin, ofloxacin, tetracycline, and / or trimethoprim);
Staphylococcushominis (including strains insensitive to methicillin, erythromycin, tetracycline and / or trimethroprim);
Staphylococcuswarneri (including strains, insensitive to erythromycin);
Streptococcusmitis (including strains that are insensitive to penicillin, erythromycin, tetracycline, and / or trimethoprim);
Streptococcus pneumoniae (including strains that are insensitive to penicillin, gentamicin, erythromycin, tetracycline, and / or trimethoprim);
Streptococcus group of viridians (including strains, insensitive to penicillin, erythromycin, tetracycline and / or trimethoprim);
Gram-negative bacteria:
Acinetobacterlwoffii, Haemophilusinfluenzae (including strains, insensitive to ampicillin); Haemophilusparainfluenzae; Klebsiellaspp.
Other microorganisms:
Chlamydiatrachomatis
Moxifloxacin acts invitro against most of the below listed microorganisms, but the clinical significance of this data is not known:
Gram-positive bacteria:
Listeriamonocytogenes, Staphylococcusaparasticus, Streptococcusagalactiae, Streptococcusmitis, Streptococcuspyogenes, Streptococcus groupC, G, F;
Gram-negative bacteria
Acinetobacterbaumannii, Acinetobactercalcoaceticus, Citrobacterfreundii, Citrobacterkoseri, Enterobacteraerogenes, Enterobactercloacae, Escherichiacoli, Klebsiellaoxytoca, Klebsiellapneumonia, Moraxellacatarrhalis, Morganellamorganii, Neisseriagonorrhoeae, Proteusmirabilis, Proteusvulgaris, Pseudomonasstutzeri;
Anaerobic microorganisms
Clostridium perfringens, Fusobacterium spp., Prevotella spp., Propionibacterium acnes
Other microorganisms
Chlamydia pneumoniae, Legionella pneumophila, Mycobacterium avium, Mycobacterium marinum, Mycoplasma pneumoniae.
There is no data on the relationship between clinical bacteriological collection of infectious diseases of the organ of vision against the background of moxifloxacin therapy. According to the epidemiological data of the European Committee for the Determination of Antimicrobial Susceptibility, the threshold values of the inhibitory concentration of moxifloxacin for various microorganisms are as follows:
Corynebacterium n / a
Staphylococcusaureus 0.25 mg / l
Staphylococcus, coag-neg. 0.25 mg / l
Streptococcus pneumoniae 0.5 mg / l
Streptococcus pyogenes 0.5 mg / l
Streptococcus, viridansgroup 0.5 mg / l
Enterobacterspp. 0.25 mg / l
Haemophilusinfluenzae 0.125 mg / l
Klebsiellaspp. 0.25 mg / l
Moraxella catarrhalis 0.25 mg / l
Morganellamorganii 0.25 mg / l
Neisseria gonorrhoeae 0.032 mg / l
Pseudomonas aeruginosa 4 mg / l
Serratiamarcescens 1 mg / l
Pharmacokinetics
When topical application occurs systemic absorption of moxifloxacin. The concentration of moxifloxacin in plasma was determined in 21 male and female patients who received moxifloxacin in the form of a drop of ocular drops in both eyes 1 drop 3 times a day for 4 days. The mean maximum concentration (Cmax) of moxifloxacin in plasma in the equilibrium state was 2.7 ng / ml, the area under the concentration-time curve (AUC) was 41.9ng h / ml. These values are approximately 1600 and 1200 times less than the mean Cmax and AUC after oral administration of the therapeutic dose of moxifloxacin 400 mg. The half-life (T1) of moxifloxacin is about 13 hours.
Indications for use
Bacterial conjunctivitis caused by microflora-sensitive moxifloxacin
Contraindications for Maxiflox
Individual hypersensitivity to the components of Maxiflox; hypersensitivity to drugs quinolone, children under 1 year.
Application in pregnancy and during breastfeeding
Pregnancy and the period of breastfeeding
There is no sufficient experience on the use of Maxiflox during pregnancy and during breastfeeding. The use of the drug during pregnancy and during breastfeeding is possible if the expected therapeutic effect exceeds the potential risk to the fetus and the baby.
Studies in animals have shown that after oral administration of moxifloxacin with breast milk, small amounts of substance are excreted. Nevertheless, if the therapeutic doses of Maxiflox are observed, development of unwanted reactions in infants is not expected.
Teratogenicity
In pre-clinical studies in animals, moxifloxacin did not have a teratogenic effect at doses of 500 mg / kg / day (approximately 21,700 times higher than the recommended daily dose for humans), but there was some decrease in fetal weight and a delay in the development of the musculoskeletal system. Against the background of a dose of 100 mg / kg / day there was an increase in the frequency of decrease in the growth of newborns.
Fertility
Studies on the effect of moxifloxacin on fertility when applied as instillations have not been conducted.
Dosing and Administration
When administering treatment, it is necessary to take into account the official recommendations for antibiotic therapy.
Only for local ophthalmic use. Not intended for use as subconjunctival injections or for administration to the anterior chamber of the eye.
Application in adults (including in elderly patients over 65 years of age)
1 drop 3 times a day in the affected eye. Improvement of the condition occurs after 5 days of therapy, but treatment should continue for another 2-3 days. In the absence of therapeutic effect after 5 days of therapy, it is recommended to revise the diagnosis and choice of therapeutic tactics. The length of the course of therapy depends on the severity of the patient's condition, clinical and bacteriological features of the infection process.
Use in the pediatric population
It is not necessary to correct the dosage regimen when used in children.
Liver and kidney failure
Correction of the dose is not required.
To prevent microbial contamination of the tip of the vial and Maxiflox, it is necessary to avoid contact with eyelids, the skin of the peri-ocular area and other surfaces.
In order to prevent absorption of Maxiflox through the nasal mucosa, it is necessary to pinch the nasolacrimal canal with a finger for 2-3 minutes after instillation.
When using several drugs for topical application in ophthalmology, the interval between their use should be at least 5 minutes, ophthalmic ointments should be used last.
Side effects of Maxiflox
General information on the safety profile of moxifloxacin in the dosage form of eye drops
In clinical trials of the dosage form of moxifloxacin for use in ophthalmology, 2252 patients received the study drug 1 drop, up to 8 times a day, 1900 of which received moxifloxacin in a 1 drop dose 3 times a day. The population for safety assessment included 1,389 patients in the US and Canada, 586 patients in Japan and 277 patients in India. According to clinical studies, no serious adverse events were reported from either the organ of vision or the body as a whole. The most common adverse reactions associated with treatment were eye irritation and pain in the eye, the total incidence of these events ranged from 1 to 2%. In 96% of patients, the severity of these reactions was mild, while in one of the patients who participated in the study, the severity of the adverse event led to the completion of the study.
Description of individual adverse reactions
There are reports of ruptures of the tendons of the shoulder joint, the joints of the hands, the Achilles tendon, and other tendons that led to a long period of incapacity for work or required surgical intervention. These phenomena were noted in patients who received systemic therapy with fluoroquinolones. According to clinical research and post-registration application, the risk of rupture of tendons in the receipt of systemic fluoroquinolone therapy may increase with the inclusion of corticosteroids in the regimen, elderly patients are at a particular risk. Most of the damage affects the tendons of the supporting joints, including the Achilles tendon.
Use Maxiflox in children
In clinical trials involving children, including newborns, a safety profile of moxifloxacin similar to the adult population was demonstrated in the form of instillations. In patients younger than 18 years, the most common pain in the eye and eye irritation, the frequency of occurrence was about 0.9%. According to the results of clinical studies in the pediatric population, there were no differences from the adult population in the profile of undesirable phenomena and their severity.
Overdose with Maxiflox
Due to the small capacity of the conjunctival cavity, there is practically no possibility of developing a local overdose with the use of drugs in the form of instillations. The total content of moxifloxacin in the preparation is too low for the development of undesirable phenomena with accidental ingestion of the contents of the vial.
Interaction Maxiflox with other drugs
Special studies of the interaction of MAXIFLOX with other drugs have not been conducted. Due to low systemic concentration after topical application in the form of instillations, interaction with other drugs is unlikely.
special instructions for Maxiflox
Patients using systemic quinolone-based drugs may experience severe, in some cases, fatal reactions of hypersensitivity (anaphylaxis), in some cases after taking the first dose. Some reactions were accompanied by collapse, loss of consciousness, Quinck's edema (including swelling of the larynx, pharynx or face), airway obstruction, dyspnoea, urticaria and skin itching.
If the allergic reaction to Maxiflox develops, Maxiflox should be discontinued. Severe acute hypersensitivity reactions to moxifloxacin and other components of the drug may require immediate resuscitation: according to indications, oxygen therapy with airway patency control can be performed.
With prolonged use of the drug, excessive growth of non-susceptible microorganisms, including fungi, can occur. In case of superinfection, it is necessary to cancel the drug and prescribe the appropriate therapy.
Inflammation and rupture of tendons were noted in systemic use of fluoroquinolones, mainly in elderly patients, as well as in patients receiving corticosteroids along with fluoroquinolones. Although the systemic concentrations of moxifloxacin after topical application in ophthalmology are significantly lower than those for oral administration, the drug should be discontinued when the first signs of inflammation of the tendons appear.
There is insufficient data to make a conclusion about the efficacy and safety of Maxiflox in the treatment of bacterial conjunctivitis in newborns, and therefore, the use of this age group in patients is not recommended. Maxiflox is not recommended for the prophylactic use or therapy of exjuvantibus (empirical treatment) of gonococcal conjunctivitis, including in the therapy of ophthalmia of newborn gonococcal etiology, due to the presence of a large number of strains resistant to moxifloxacin Neisseriagonorrhoeae. Patients with infectious diseases of the eye caused by Neisseriagonorrhoeae should receive appropriate systemic therapy.
It is not recommended to use Maxiflox in the treatment of infectious diseases of the organ of vision caused by Chlamydiatrachomatis in patients younger than 2 years, since there is no information on the study of the drug in this category of patients. The use of Maxiflox in patients older than 2 years with eye diseases caused by Chlamydiatrachomatis should be combined with systemic therapy.
Patients should receive appropriate treatment for ophthalmia of newborns; so, with the development of conjunctivitis of chlamydial and gonorrheal etiology, this type of treatment will be systemic therapy.
In the presence of infectious diseases of the anterior segment of the eyeball, contact lenses are not recommended.
Impact on the ability to drive vehicles and mechanisms
As in the case of instillations of other drugs, a temporary blurring of vision after application of the drug is possible. Before the restoration of clarity of visual perception is not recommended to drive and other mechanisms.
Form of issue
Eye drops 5 mg / ml.
5 ml of the drug are placed in a 5 ml polyethylene bottle, closed with a stopper and a lid with a safety ring made of polyethylene. A label is attached to the vial.
One bottle together with the instruction for use is placed in a cardboard box.
Storage conditions
At a temperature of no higher than 25 ° C, in the original packaging.
Keep out of the reach of children.
Shelf life - 3 years.
After opening the bottle, the drops should be used within 4 weeks.
Do not use after the expiration date!
Leave conditions
To buy Maxiflox the prescription is not required.