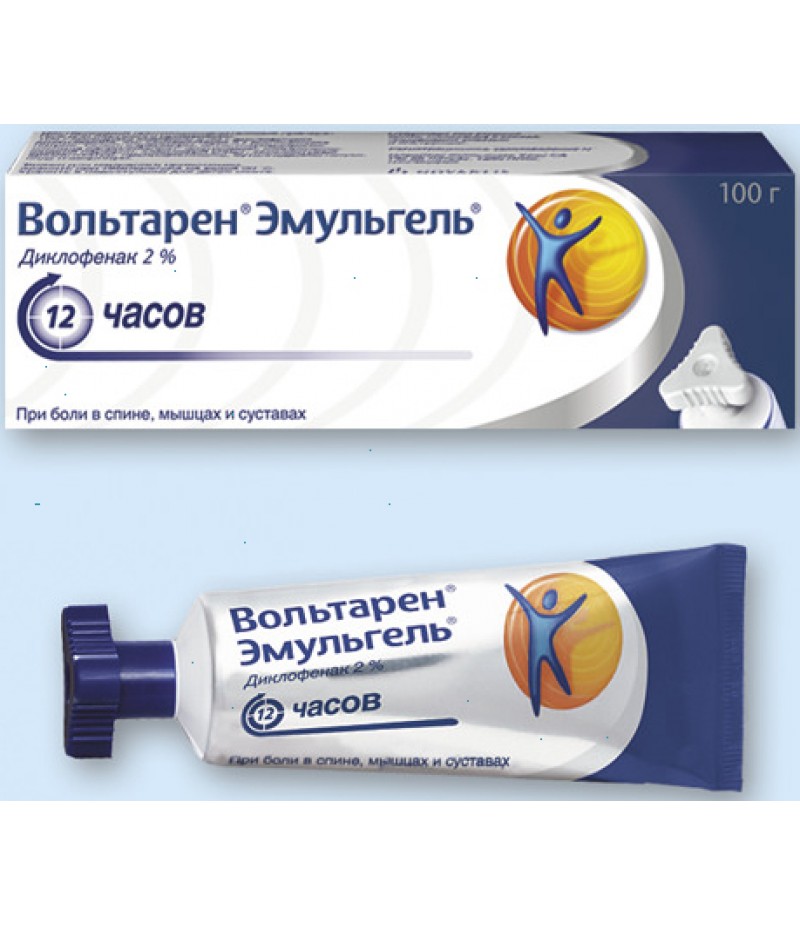
Voltaren emulgel 1% tube 100g
- $25.28
- 3 or more $23.90
- Availability:In Stock
Voltaren emulgel instruction for useYou can buy Voltaren emulgel on this pageClinical and pharmacological groupNSAIDs for external usepharmachologic effectNSAIDs for external use. Diclofenac has a pronounced analgesic and anti-inf..
Tags: gel
Voltaren emulgel instruction for use
You can buy Voltaren emulgel on this page
Clinical and pharmacological group
NSAIDs for external use
pharmachologic effect
NSAIDs for external use. Diclofenac has a pronounced analgesic and anti-inflammatory and antipyretic effect. By indiscriminately inhibiting COX-1 and COX-2, it breaks the metabolism of arachidonic acid. Voltaren Emulgel is used to eliminate pain syndrome and inflammation in the joints, muscles and ligaments of traumatic or rheumatic origin, contributing to the reduction of pain and swelling associated with the inflammatory process, increasing the mobility of the joints.
Pharmacokinetics
Suction
The amount of diclofenac absorbed through the skin is proportional to the area of the treated surface and depends on both the total dose of the drug applied and the degree of hydration of the skin. After applying Voltaren Emulgel, a gel for external application of 2% (2 applications per day) to the surface of the skin with an area of 400 cm2, the concentration of the active substance in the plasma corresponds to its concentration when using 1% diclofenac gel (4 applications per day). On the 7th day, the relative bioavailability of the drug (AUC ratio) is 4.5% (for an equivalent dose of the sodium salt of diclofenac). When wearing a moisture-permeable bandage, the absorption did not change.
Distribution
When applying the drug on the affected joint, the concentration of diclofenac in the plasma, synovial membrane and synovial fluid was determined. Cmax in plasma was approximately 100 times lower than after oral administration of the same amount of diclofenac.
The binding of diclofenac to plasma proteins is 99.7%, mainly with albumin (99.4%).
Diclofenac is predominantly distributed and retained deep in tissues prone to inflammation, such as joints, where its concentration is 20 times higher than in plasma.
Metabolism
The metabolism of diclofenac is partly due to glucuronization of the unmodified molecule, but mainly through single and multiple hydroxylation, which leads to the formation of several phenolic metabolites, most of which are converted into glucuronide conjugates. Two phenolic metabolites are biologically active, but to a much lesser extent than diclofenac.
Excretion
The total systemic plasma clearance of diclofenac is 263 ± 56 ml / min.
The final T1 / 2 is 1-2 h. The T1 / 2 metabolites, including the two pharmacologically active metabolites, are also short-lived and are 1-3 h. One of the metabolites (3'-hydroxy-4'-methoxydiclofenac) has a longer T1 / 2, but this metabolite is completely inactive. Most of the diclofenac and its metabolites are excreted in the urine.
Indications
- pain in the back with inflammatory and degenerative diseases of the spine (sciatica, osteoarthrosis, lumbago, sciatica);
- pain in the joints (including joints of the fingers, knees) with rheumatoid arthritis, osteoarthrosis;
- pain in the muscles (due to stretching, overstrain, bruises, injuries);
- inflammation and puffiness of soft tissues and joints due to injuries and rheumatic diseases (tendovaginitis, bursitis, periarticular tissue lesions, wrist syndrome).
Contraindications
- hypersensitivity to diclofenac or other components of the drug;
- propensity to occurrence of attacks of bronchial asthma, skin rashes or acute rhinitis with the use of acetylsalicylic acid or other NSAIDs;
- violation of the integrity of the skin in the intended site of application;
- III trimester of pregnancy;
- the period of lactation (breastfeeding);
- Children under 12 years old.
With caution should prescribe the drug with hepatic porphyria (in the phase of exacerbation), erosive and ulcerative lesions of the gastrointestinal tract, severe violations of liver and kidney function, chronic heart failure, bronchial asthma, and in the first and second trimesters of pregnancy and elderly patients.
Dosage
The drug is used externally.
Adults and children over 12 years of age, the drug is applied to the skin 2 times / day (every 12 hours: preferably in the morning and in the evening), lightly rubbing into the skin.
The required amount of the drug depends on the size of the painful area. A single dose of the drug - 2-4 g (which is comparable in volume to the size of a cherry or walnut, respectively) is enough for processing a zone of 400-800 cm2.
If the hands are not a zone of pain localization, then after applying the drug, they must be washed.
The duration of treatment depends on the indications and the effect observed. The gel should not be used for more than 14 days with post-traumatic inflammations and rheumatic diseases of soft tissues without a doctor's recommendation. If the therapeutic effect is not observed after 7 days or the condition worsens, the patient should consult a doctor.
To remove the protective membrane, use a screw cap as a key (a recess with protrusions on the outside of the cover). Align the recess on the outside of the lid with the tubular protective membrane and turn. The membrane must separate from the tube.
Side effects
Unwanted reactions are mainly characterized by moderately expressed and transmitted skin manifestations at the site of application of the gel. In very rare cases, the development of allergic reactions is possible.
Determination of incidence of adverse reactions: very often (> 10%), often (≥1%, but ≤10%), infrequently (≥0.1%, but ≤1%), rarely (≥0.01%, but ≤0.1%); very rarely (≤0.01%).
Infectious and parasitic diseases: very rarely - pustular eruptions.
On the part of the immune system: very rarely - generalized skin rash, allergic reactions (urticaria, hypersensitivity: angioedema).
From the respiratory system: very rarely - attacks of suffocation, bronchospastic reactions.
From the skin: often - erythema, dermatitis, incl. contact dermatitis (symptoms: eczema, itching, swelling of the treated area of the skin, rash, papules, vesicles, peeling); rarely bullous dermatitis; very rarely photosensitization reactions.
Overdose
Due to low systemic absorption during the application of the gel, an overdose is unlikely.
Symptoms: with accidental ingestion, it is possible to develop systemic adverse reactions.
Treatment of an overdose with accidental ingestion: gastric lavage, induction of vomiting, activated charcoal, symptomatic therapy. Dialysis and forced diuresis are not effective due to the high degree of binding of diclofenac to plasma proteins (about 99%).
Drug Interactions
Voltaren Emulgel can enhance the effect of drugs that cause photosensitivity.
Clinically significant interaction with other drugs is not described.
special instructions
Voltaren Emulgel should only be applied to intact skin, avoiding exposure to open wounds.
After applying the drug, a bandage bandage is allowed, but no airtight occlusive dressings should be applied. In the case of development after application of the skin rash, its use should be discontinued.
Do not allow the drug to enter the eyes and mucous membranes.
The drug contains propylene glycol, which in some people can cause mild local irritation. It also contains butylhydroxytoluene, which can cause local skin reactions (eg, contact dermatitis) or eye and mucous membrane irritation.
Impact on the ability to drive vehicles and manage mechanisms
Does not affect.
Pregnancy and lactemia
Due to the lack of data on the use of Voltaren Emulgel during pregnancy, the use of the drug in the first and second trimesters of pregnancy is recommended only for the doctor's prescription, comparing the benefits to the mother and the risk to the fetus.
The drug is contraindicated in the third trimester of pregnancy in connection with the possibility of lowering the tone of the uterus and / or premature closure of the arterial duct of the fetus.
In connection with the lack of data on the isolation of the active substance Voltaren Emulgel with breast milk, the drug is not recommended for use during breastfeeding. If nevertheless it is necessary to use the drug, it should not be applied to the mammary glands or to a large surface of the skin and not to be used for a long time.
Application in childhood
Contraindicated: children under 12 years.
In case of violations of kidney function
With caution appoint for severe violations of kidney function.
With violations of liver function
With caution appoint for severe violations of the liver.
Application in old age
Caution should be given to elderly patients.
Conditions of leave from pharmacies
The drug is dispensed without a prescription.
Terms and conditions of storage
The drug should be stored out of reach of children at a temperature of no higher than 30 ° C. Shelf life - 3 years.